Read More
Wallis WangUnder the new "1+" drug registration mechanism, the approval from one drug regulatory authority - instead of the two previously required - is enough for new drugs for life-threatening diseases recognized by local experts.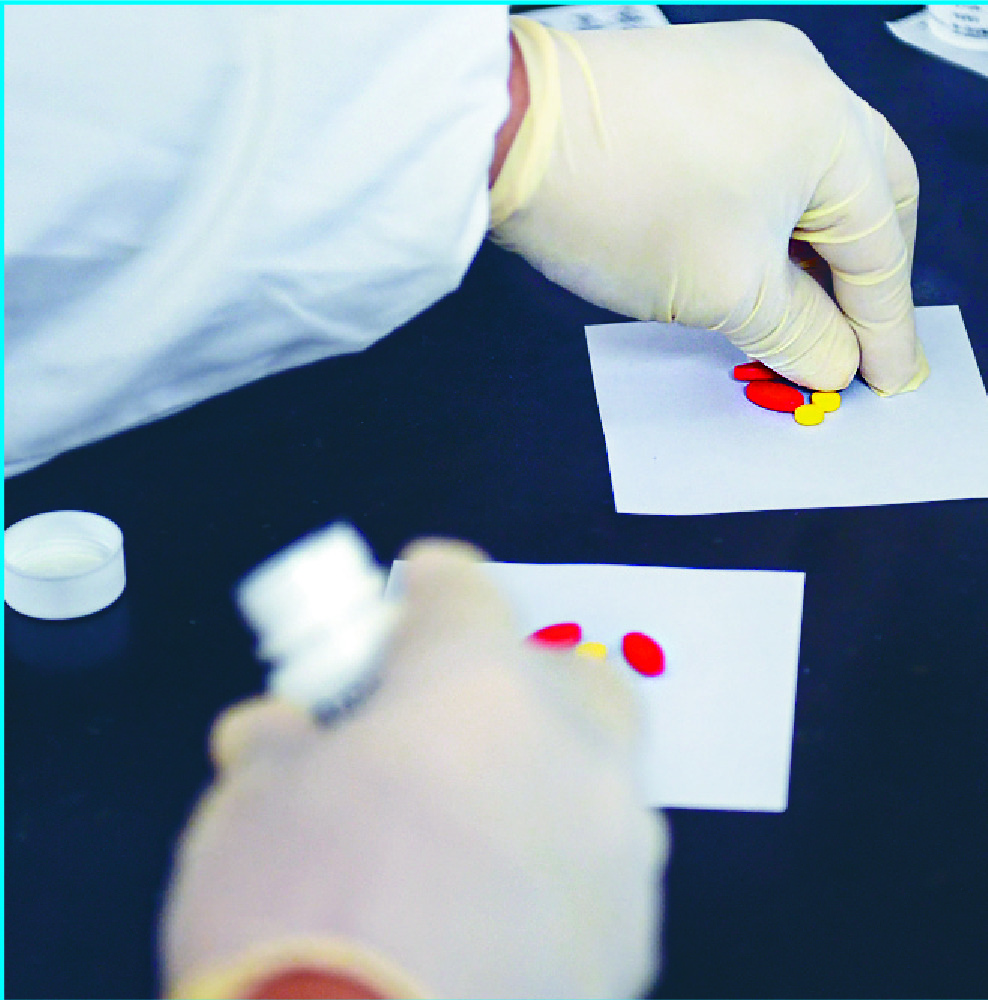
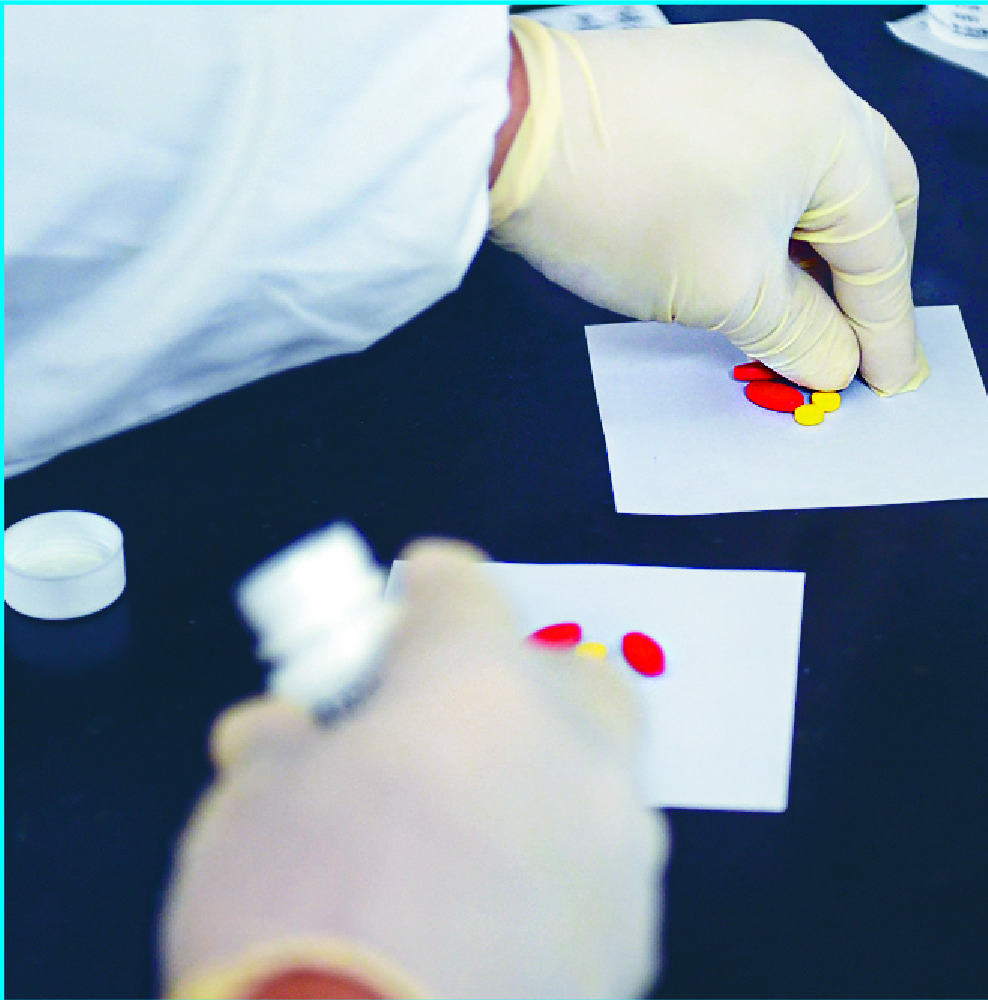
Two mainland drugs for colorectal cancer are now available in the SAR after Hong Kong authorities approved their use under a simplified drug registration mechanism that came into effect in November.
ADVERTISEMENT
SCROLL TO CONTINUE WITH CONTENT
In a document submitted to the Legislative Council panel, the Department of Health said it received 126 inquiries from 55 pharmaceutical companies as of February 13 since the "1+" mechanism came into effect.
The department hosted four online briefings on the mechanism, which were attended by 175 pharmaceutical representatives.
"Under the '1+' mechanism, we have approved two new cancer drugs from the mainland. The two drugs are oral targeted therapy drugs of different doses for colorectal cancer and can be used on patients who do not respond to or are not suitable for conventional chemotherapy," the department said.
"The first batch of the two new drugs arrived in Hong Kong in early February, bringing new hopes to patients," it added.Some pharmaceutical companies have expressed interest in new drug registration under the new mechanism, the department said.
"The '1+' mechanism will facilitate the local registration of drugs from all around the world so that patients can benefit from new medications sooner. It can also attract more drug development and clinical trials to be held in Hong Kong," it said. "Local experts' review of the drugs' clinical data and their approval will make sure all registered drugs are safe, effective and of high quality while strengthening local drug review ability and promoting talent development."Currently, Hong Kong health authorities adopt a "secondary evaluation" approach for vetting and approval of other drugs for non-critical diseases, meaning they rely on registration approvals from drug regulatory authorities in other countries.
In 2022, the government officially recognized four more drug regulatory authorities - the mainland, Brazil, South Korea and Singapore - bringing the total to 36 authorities.Pharmaceutical companies have to submit approvals from at least two out of the 36 to have drugs registered in Hong Kong.
Since the update of the recognized drug regulatory bodies list, Hong Kong has received registration applications for 12 drugs with approvals from four newly added authorities, among which eight have been approved.wallis.wang@singtaonewscorp.com
Some pharmaceutical companies have expressed interest in new drug registration under the new mechanism, the department said.