A small trial of an experimental vaccine has shown promising results for patients with the most aggressive type of breast cancer, according to a report published on Thursday in Genome Medicine.
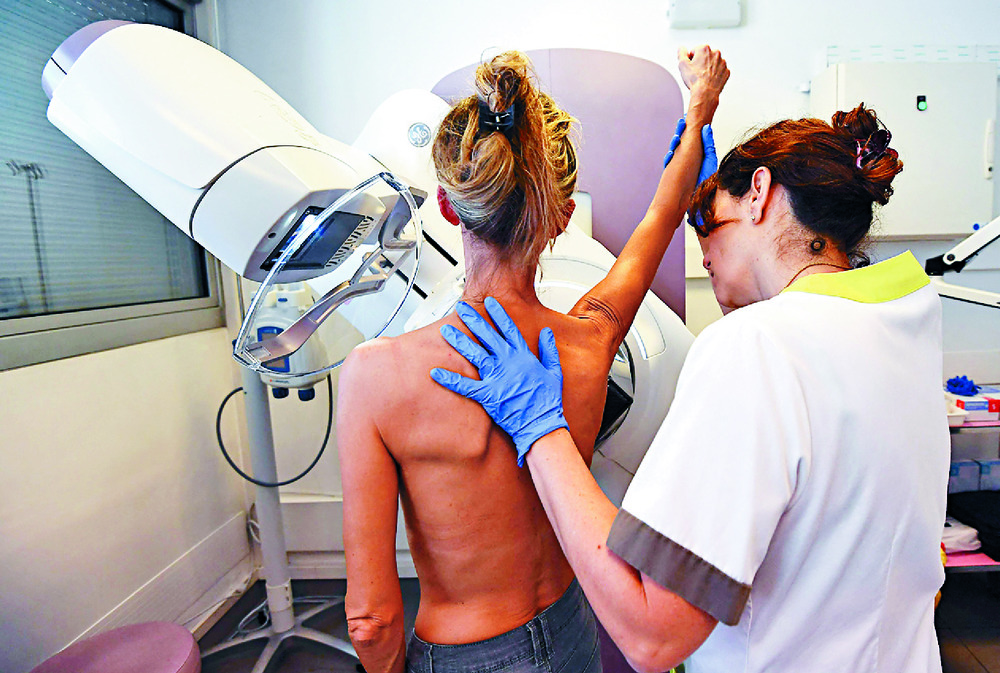
Eighteen patients newly diagnosed with triple-negative breast cancer that had not yet spread to other organs received three doses of the personalized vaccine after undergoing standard chemotherapy and surgery to remove the tumor.
Three years later, 16 remained cancer-free. The study did not include a control group, but the researchers note that with standard care alone, about half of the patients would be expected to be cancer-free at three years.
The vaccine is designed to target key gene mutations in tumors called neoantigens, new proteins that have formed in cancer cells due to mutations in the tumor DNA. The vaccines are also designed to train immune cells to recognize and attack any cells bearing these mutations.
As reported in a separate paper in the same journal, the researchers created software to identify the neoantigens most likely to trigger a strong immune response.
Small, early studies like this are designed to test safety, not effectiveness. Still, the results "were better than we expected," said William Gillanders of Washington University school of medicine in St Louis, who headed the trial. "We are continuing to pursue this vaccine strategy and have ongoing randomized controlled trials that do make a direct comparison between the standard of care plus a vaccine, versus standard of care alone. We are encouraged by what we're seeing so far."
Each patient received three doses of a personalized vaccine tailored to home in on key mutations in their specific tumor and train immune cells to recognize and attack any cells bearing these mutations.
Triple-negative breast cancer is an aggressive tumor type that grows even in the absence of the hormonal fuel that drives growth of other types of breast cancer. To date, triple-negative breast cancer has no targeted therapies and is usually treated with traditional approaches that include surgery, chemotherapy and radiation therapy.
For this trial, patients with triple-negative breast cancer who still had evidence of a tumor remaining after a first round of chemotherapy were eligible to participate. Such patients are at high risk of cancer recurrence even after the remaining tumor is surgically removed.
After surgical removal, the research team analyzed and compared the tumor tissue with the patient's healthy tissue to find unique genetic mutations. Such mutations in cancer cells alter the proteins only in the tumor, making it possible to train the immune system to go after the altered proteins and leave healthy tissues alone.
Using software they designed, the researchers selected neoantigens. On average, each patient's vaccine contained 11 neoantigens (ranging from a minimum of four to a maximum of 20) specific to their tumor.
One of the goals is to make these computational resources widely accessible to researchers and clinicians worldwide.
(By Nancy Lapid and staff reporter)