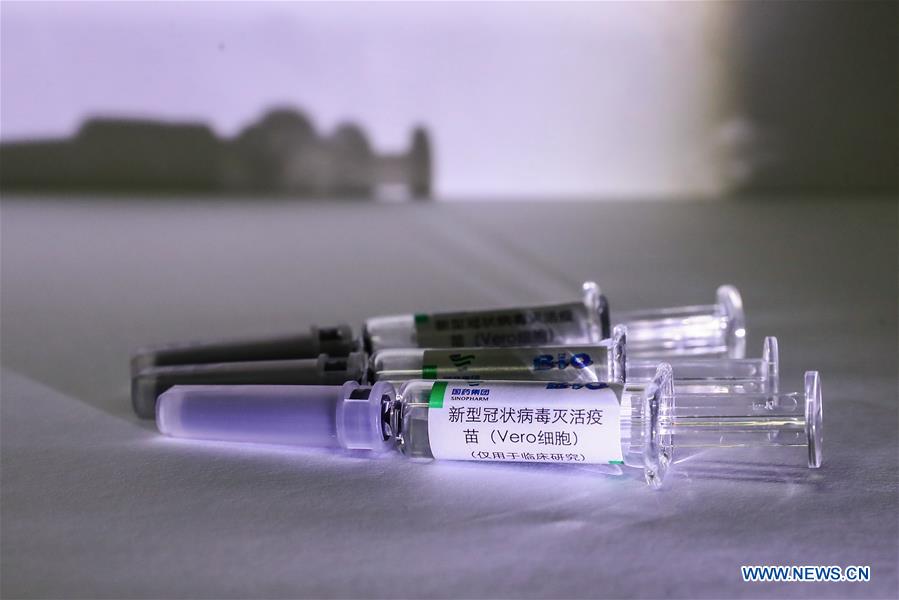
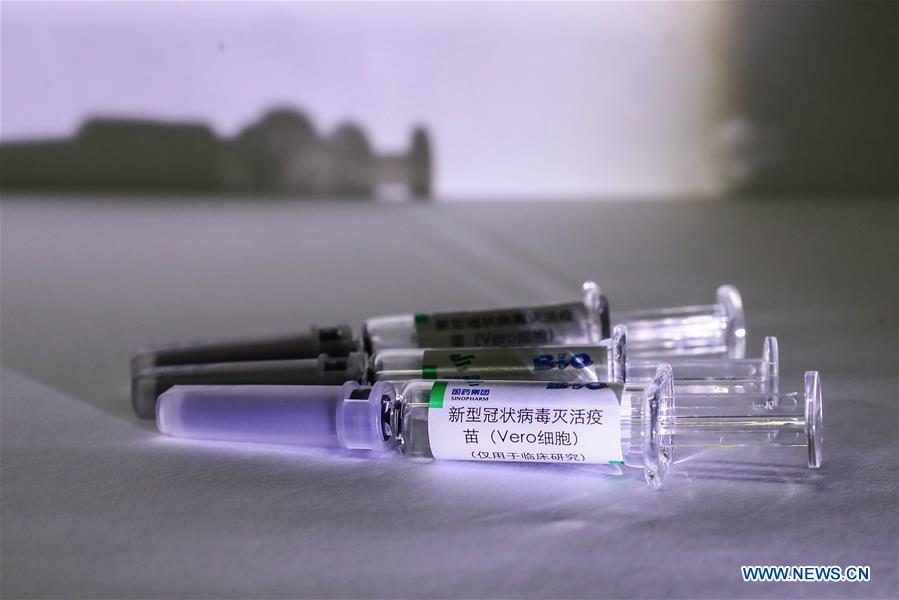
China has approved two coronavirus inactivated vaccine candidates for clinical trials, according to the State Council joint prevention and control mechanism against the coronavirus today, Xinhua reports.
The two vaccine candidates are developed by Wuhan Institute of Biological Products under the China National Pharmaceutical Group (Sinopharm) and Sinovac Research and Development Co., a company based in Beijing. Clinical trials of the two vaccines have started.
Samples of the coronavirus inactivated vaccine are seen at a production plant of China National Pharmaceutical Group (Sinopharm) in Beijing, on April 10, 2020.