Read More
German biotech company BioNTech will be supplying Covid-19 vaccines to Hong Kong from Europe in collaboration with mainland firm Fosun Pharma, instead of US-Swiss giant Pfizer.
ADVERTISEMENT
SCROLL TO CONTINUE WITH CONTENT
Britain became the first Western country to start vaccinating people with an approved vaccine last Tuesday after it granted emergency use authorization for the Pfizer-BioNTech vaccine, made with the new technology mRNA.
When Chief Executive Carrie Lam Cheng Yuet-ngor on Friday announced procuring a maximum of 7.5 million doses of vaccine developed by BioNTech in collaboration with mainland biopharmaceutical company Fosun Pharma, she first mentioned Pfizer as a co-supplier.
But in a statement that night, the government said Pfizer is BioNTech's partner for vaccines in regions outside China and that the shots to be supplied to Hong Kong will only be in collaboration with Fosun Pharma.
Pfizer's absence sparked speculation that the BioNTech shots will be manufactured on the mainland, unlike those supplied to the United States, which are made in Pfizer plants in Michigan and Wisconsin.
On Saturday night, the government clarified that the shots to be supplied to Hong Kong will be manufactured in Europe and that the first batch of one million doses is expected to be delivered in the first quarter at the earliest.
While Fosun Pharma is responsible for clinical trials, regulatory applications, sales and marketing of the jabs in China, the injections will be provided by BioNTech.
BioNTech has told The Standard that its partnership with Pfizer is worldwide, except in China.
The German company partnered with the US-Swiss giant Pfizer in research and development of the mRNA-based vaccine, which was funded by the German government.
The production of mRNA vaccines requires specialized know-how, in which BioNTech has developed significant expertise in the field over the past 12 years.
As for the BioNTech-Fosun collaboration, it focuses on the development, manufacture and distribution of BioNTech's mRNA-based vaccine in Greater China, which includes mainland, Hong Kong, Macau and Taiwan.
Producing the vaccines in Europe leads to another question on logistics, as the 95 percent effective BioNTech vaccines must be transported and stored in ultra-cold cargoes of minus-70 degrees Celsius, to which authorities said Fosun has entered discussion with logistics and cold-storage companies, as well as the Airport Authority on necessary arrangements.
Chief Secretary for Administration Matthew Cheung Kin-chung said on his blog the government has started an emergency legislation to accelerate local registration of vaccines for administration schemes to roll out as early as possible.
To get enough vaccines to cover the whole population, the government has secured another 7.5 million shots from Sinovac (Hong Kong) Ltd. They will be produced in Beijing and the first batch of one million will arrive next month at the earliest.
The third vaccine for Hongkongers will be jointly developed by AstraZeneca and the University of Oxford.
Jane Cheung, Mary Ann Benitez and Maisy Mok
Editorial: Vaccine backtrack a PR backfire
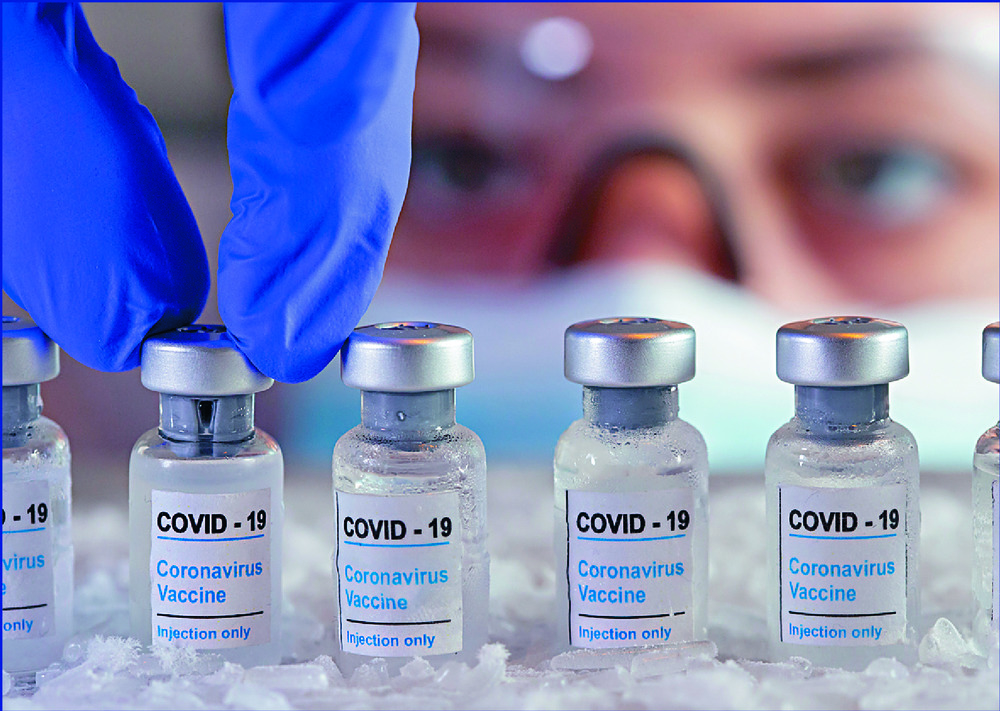
The 7.5 million vaccines to be shipped to Hong Kong will be produced in Europe. REUTERS

Fosun Pharma is BioNTech's partner in Greater China.REUTERS