The first chance for Americans to see how strong the evidence really is about the Pfizer and BioNTech coronavirus vaccine will come in early December at a public meeting of the US Food and Drug Administration scientific advisers.
So far, what is known is based only on statements from Pfizer and BioNTech.
Of 170 infections detected to date, only eight were among people who had received the actual vaccine and the rest had got a dummy shot.
On the safety side, the companies cite results from 38,000 study participants who’ve been tracked for two months after their second dose. That’s a milestone FDA set because historically, vaccine side effects don’t crop up later than that.
“We’ll drill down on these data,” said FDA adviser Dr. Paul Offit of the Children’s Hospital of Philadelphia.
Think of it like science on trial. A few days before the meeting, the FDA will release its own internal analysis. That sets the stage for the advisers’ daylong debate about any signs of safety concerns and how the new vaccine technology works before rendering a verdict.
They will recommend not just whether FDA should allow broader use of the vaccine generally but if so, for whom. For example, is there enough proof the vaccine works as well for older, sicker adults as for younger, healthier people?
There is still no guarantee. “We don’t know what that vote’s going to be,” said former FDA vaccine chief Norman Baylor.-AP
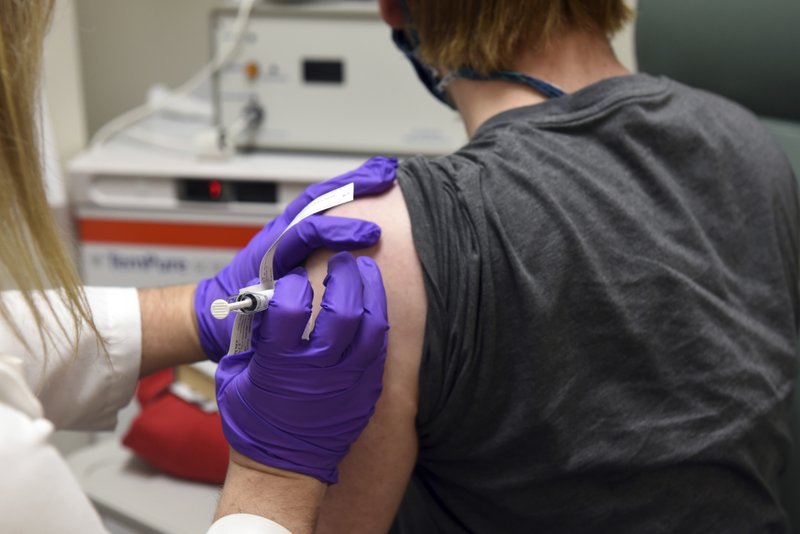
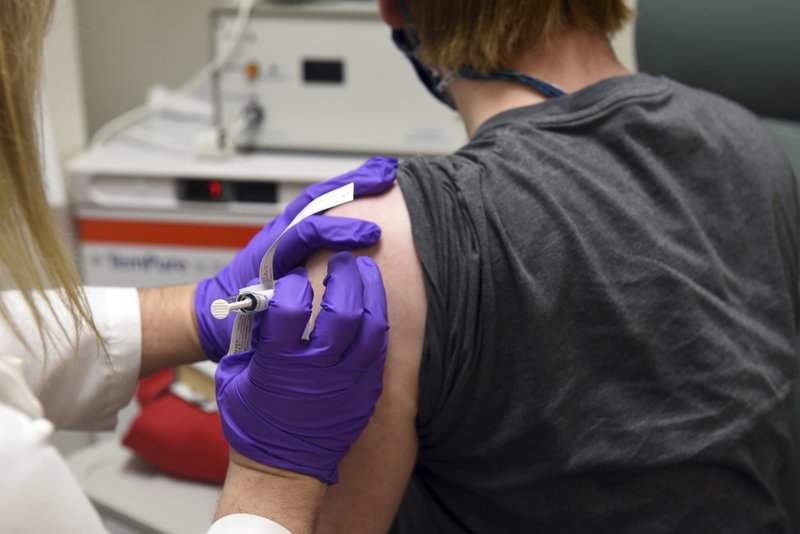
This May 4, file photo provided by the University of Maryland School of Medicine, shows the first patient enrolled in Pfizer's coronavirus vaccine clinical trial at the University of Maryland School of Medicine in Baltimore.